With only 10 months to go, the world is gearing toward action in the light of the possible Brexit scenarios. At this time where first announcements of potential withdrawal from UK are being made by industries that rely on just-in-time delivery of parts to keep the costs down (e.g. airline, automotive), the authorities are shifting gears as well. That is obvious when reading the EMA guidance to industry in relation to Brexit updated last week.
On its Brexit pages, EMA indicates it will start operations in Amsterdam on March 30, 2019, at the latest. But in contrast to the device world that is not yet seeing formal guidance on Brexit, EMA provides formal guidance to pharma industry on how the transition from MHRA as rapporteur or co-rapporteur will take place. The EMA guidance clearly specifies its considerations to be aligned with a hard Brexit as worse case scenario: “This means that unless a ratified withdrawal agreement establishes another date, all Union primary, and secondary law ceases to apply to the UK from 30 March 2019, 00:00h (CET) and the UK will then become a 'third country'.”
At this stage the EU27 Member States and EMA completed the redistribution of the MHRA's portfolio of almost 400 centrally authorized products to rapporteurs and co-rapporteurs from the EU27 plus Iceland and Norway (EU27+2), in preparation for Brexit. Late April 2018, EMA informed the relevant manufacturers of the new (co)-rapporteurs. Structured ‘knowledge transfer packs’ will enable the transfer as of September 2018. The new (co-)rapporteurs will only take full responsibility for these products as of 30 March 2019.
Interestingly, this massive operation only focusses on the responsibilities in the post-authorisation stage in a medicine’s lifecycle. As the CHMP approvals are basically an approval from all members based on a recommendation by 2 members, the conclusion seems to be that pre-market approval and historic post-market oversight remain valid, and ownership for continuation will transfer.
With this step, formally part of the second stage in the business continuity plan, EMA steps away from the ‘business as usual’ path that was still in place until now.
The Q&A document that is part of the Brexit-related guidance for manufacturers contains some sections on UK-notified bodies as well. It emphasizes the black and white wording of the Commission Notice on the withdrawal of the United Kingdom from the EU, that identifies UK Notified Body certificates will lose validity on March 30, 2019 (unless an agreement changes this).
It further specifies the impact on previously held consultations from EMA to Notified Bodies on e.g. human blood derivatives and biotech medicines, indicating that as of Brexit day, former UK notified bodies may no longer apply for such consultations.
Another guidance document in the Brexit section of EMA informs manufacturers that for pharmaceuticals that co-pack medical devices that are CE marked by a UK notified body, they need to change notified body, or replace the device with an equivalent medical device, and file a type IACN notification. Such changes need to be notified immediately after implementation, and actual implementation of such changes must in any case take place before 30 March 2019, irrespective of the variation type.
With these guidance, the impact of Brexit on medical devices in device/drug combinations and in drug/device combinations have become much clearer indeed, as indicated in below summary table:

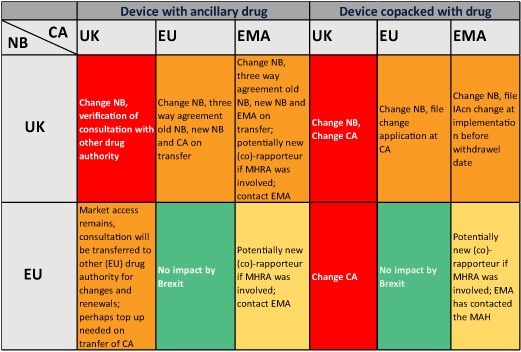
Unexpectedly, the EMA guidance seems to suggest that the historic consultation processes might remain valid, as they only restrict new procedures. That might imply that the historic Notified Body consultations performed by MHRA might be transferred to other EU27+2 agencies, without having to be re-done.
Needed at this stage is guidance and expectations from EU27 Notified bodies, and from EU27+2 drug agencies on their acceptance of work previously done by UK notified bodies and by UK MHRA for the medical devices with pharmaceuticals providing ancillary action.
Need help with your combination products or with the EU-MDR implementation, feel free to get our team in to support you.
Gert